A drinking bird consists of two glass bulbs, joined by a tube. The tube extends nearly all
the way into the bottom bulb but does not extend into the top bulb. The bottom bulb, the body, contains a liquid, mostly
methylene chloride or ether.
The top bulb, the head, is covered by a felt like material.
There is no air inside the bird, so the space inside the is filled with vapor.
To get the bird started the head must be made wet by water. Then the thermodynamic cycle begins:
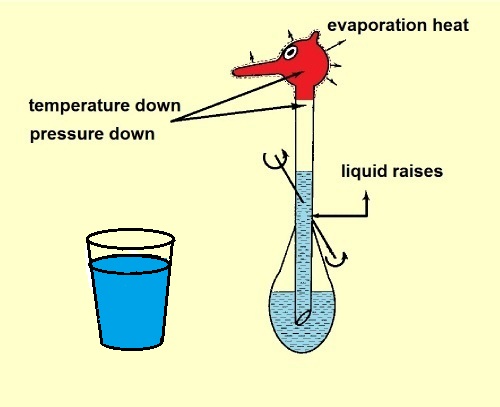
Because of the evaporation of the water in the filt of the head, the temperature of the heads drops. (Some vapor inside the bird will condensate
and that will reduce a little the temperature decrease.) The pressure inside the head drops and the liquid will be pushed upwards.
The bird becomes top-heavy and it dunks its head into the cup with water for a "drink".
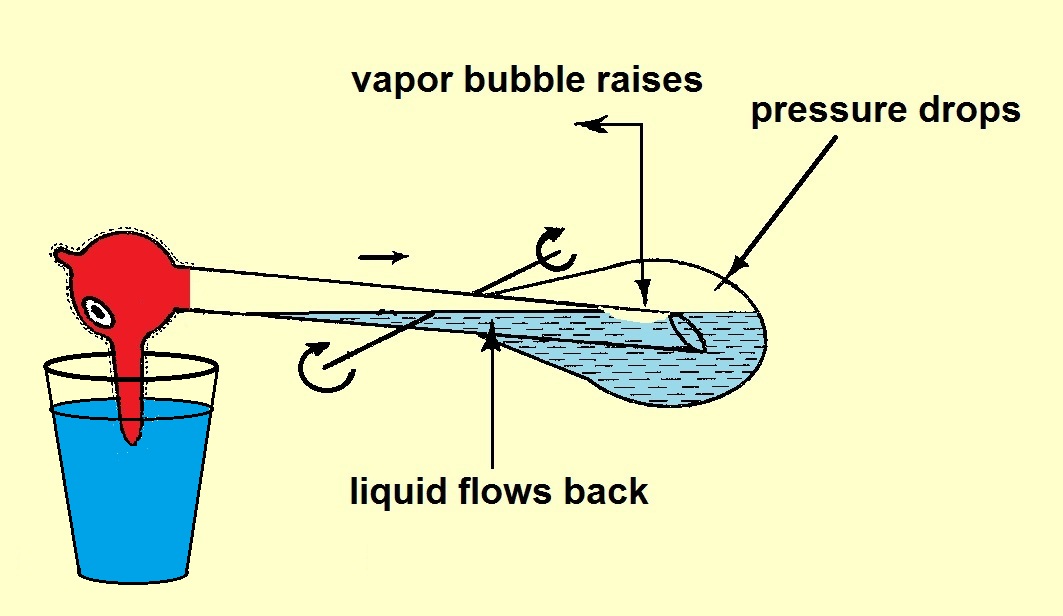
When the bird is in (almost) horizontal position the liquid in the tube flows back into the body. At the same time vapor from the body moves up
into the tube. The center of mass becomes below the turning point and the bird moves back.
The bird doesn't realy drink, but due to dipping in the water the head remains wet. Under room conditions the cycle will repeat endless unless
there in no more water in the cup or when the humity of the environment is high. When the bird and the cup are put under a bell jar
the relative humidity of the air around the bird will raise to 100%, there will be no more evaporation of water on the head and the bird will stop.
Physical aspects of the drinking bird:
- capilarry action
- combined gas law: pV=nRT
- evaporation heat
- condensation heat
Click here to see the original drawing that is part of the patent 2,402,463 from M.V. Sullivan.
The drawings are taken from "Het duikende eendje" by A. Ziggelaar.
